
Image based on sonochemical reactions (luminol) inside a BuBble bag.
BuBble bags can be used for other purposes than just cleaning. In labs, ultrasonic baths are often used for chemical and biochemical processes. Since these processes rely on cavitation (partly because of the OH radicals that are generated), you can enhance the processes by using our BuBble bags, since these bags generated more cavitation bubbles.
Both the yield and the reproducibility of (bio)chemical reactions is improved, which is an important advantage for e.g. sonochemistry.
Below is a list of several relevant (bio)chemical processes that can be performed/enhanced with ultrasound.
Some processes have been detailed below; click on the corresponding links.
- Emulsification
- Homogenisation
- Dissolution
- Mixing
- Sonochemical reactions
- Leaching
- Particle synthese
- Crystallisation/precipitation
- Exfoliation (graphite, nanosheets)
- Extraction
- Liposome formatie
- Sono(chemi)luminescence
- Cell lysis
- DNA extraction
EMULSIFICATION
Emulsions are everywhere, both in the lab and in daily life (e.g. paint, mayonaise). These emulsions are typically made from difficult-to-mix materials such as oil and water. Oscillating bubbles generated by ultrasound are excellently suitable for adding motion to the liquid, which allows the materials to mix.
We have studied the emulsification of hexadecane in a detergent solution (SDS) inside a BuBble bag. Using a particle sizer, the resulting droplet size distribution is measurement, both for bags with and without our wall modification. Outcome: The wall modification of the BuBble bags result in a 10-fold refinement of the emulsion. This puts the technique on par with industrial emulsification technologies such as a high pressure homogeniser.
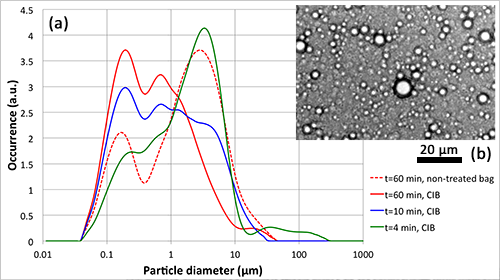
Emulsification in a BuBble bag. (a) The graph shows that the droplet size is decreasing over time and due to the BuBble bag (here named CIB-Cavitation Intensifying Bag). (b) A microscope photo of an emulsion, showing oil droplets in a detergent solution.
More results from this study will be published soon at the CAV2015 conference, December 6-10, 2015 in Lausanne, Switzerland.
Work done in collaboration with Ralph van Zwieten and Karin Schroen (Wageningen University).
SONOCHEMICAL REACTIONS
One of the effects of ultrasound cavitation is the splitting of water molecules, thereby generating radicals such as •OH. These radicals are strongly reactive and therefore can be used in various processes such as sonochemistry, cell lysis, and scavenging of contaminating molecules.
We did a pilot study on the amount of radicals produced within a BuBble bag and within an exposure time of a few minutes. We find that the micropits wall modification leads to a slight increase in amount of radicals, but perhaps more importantly also reduces the scattering in the results. This means that the process becomes more reproducible by using our BuBble bags – something that is often a problem for the sonochemistry community.
Work done in collaboration with Youlin Liu (University of Science and Technology China).
CELL LYSIS
Cavitation bubbles are capable of opening cell walls and even destroy (kill) entire cells. This is useful for applications such as DNA extraction. Typically, cell samples are treated in Eppendorf tubes or glass containers, but these significantly dampen the ultrasound pressure.
Preliminary tests with our BuBble bags have shown that our bags lead to a lot more cell lysis compared to Eppendorf tubes; the improvement over glass tubes was less prominent. More detailed results will follow soon.
Work done in collaboration with Adithya Sridhar and Severine le Gac (MIRA, University of Twente).



